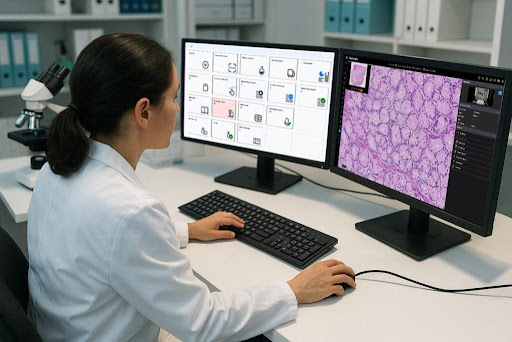
There is a phrase that circulates in laboratory medicine circles: pathology is the last analog department in a digital hospital. It is a bit of an overstatement, since plenty of pathology labs have been investing in digital tools for years, but it captures something real about the pace of digitization in pathology compared to other areas of healthcare. While radiology went digial with PACS systems decades ago, and clinical chemistry labs have had automated analyzers and information systems for years, anatomic pathology held onto its analog roots much longer than almost any other clinical discipline.
That is changing. And it is changing quickly enough that what was considered an advanced or progressive lab five years ago is starting to look like the baseline expectation today. Digital pathology, the capture of whole slide images that can be viewed, annotated, and shared electronically, has crossed a regulatory threshold that once held back adoption. The FDA’s clearance of digital pathology systems for primary diagnosis, beginning in 2017 and followed by several other vendors, removed the regulatory ambiguity that had made many labs cautious about fully committing to digital workflows.
Three Distinct Drivers of Digital Adoption
The drivers of this shift are worth understanding because they are not all the same thing. Labs are moving to digital pathology for different reasons, and understanding those reasons helps clarify what investments make sense and when.
The first group is moving primarily for operational benefits. These labs want the ability to have pathologists review cases remotely, to share difficult cases with subspecialists without physically shipping glass slides, and to retrieve archived cases without searching through physical storage. These practical benefits became particularly visible during the COVID-19 pandemic, when remote work arrangements that once seemed impractical for pathology became urgent necessities.
The second group is investing in digital pathology primarily as enabling infrastructure for AI-assisted analysis. AI algorithms for pathology image analysis require digital slides. Labs that want to leverage the growing ecosystem of AI diagnostic tools need whole slide imaging infrastructure in place first.
The third group is being pulled toward digital pathology by the changing expectations of the clinical teams they serve. Oncologists coordinating multidisciplinary tumor boards expect pathology images integrated into their case management platforms. Surgeons want to discuss margin status with real-time access to the same images the pathologist is reviewing. These expectations create pressure on pathology departments to adopt the digital infrastructure that makes this kind of integration possible.
The Central Role of the LIS Platform
The LIS platform is a critical piece of this puzzle because it sits at the center of the information flow. A lab might have excellent whole slide imaging hardware and capable AI analysis tools, but if those systems are not integrated with the information system managing the rest of the case workflow, the result is a fragmented experience. Pathologists toggle between multiple interfaces, results have to be manually reconciled, and reporting does not capture the full picture.
NovoPath’s approach to this integration challenge reflects the recognition that the LIS has to function as the connective tissue of the digital pathology lab. The platform’s integration capabilities extend to digital imaging viewers, AI analysis tools, molecular diagnostics platforms, and other lab systems, allowing the information generated at each stage of the workflow to flow into a unified case record rather than being siloed in separate systems. Learn more about NovoPath’s take on digital pathology and AI here: www.novopath.com/guides/the-practical-guide-to-operationalizing-digital-pathology-ai-in-the-lab/
Interoperability: Still an Evolving Area
Standardization in digital pathology is still an evolving area. There is a recognized need for interoperability standards that would allow images captured on one vendor’s scanner to be processed seamlessly by another vendor’s AI tools. The DICOM standard has been extended to support digital pathology images, which is an important step, but full interoperability in clinical practice remains a work in progress. Labs evaluating digital pathology platforms and LIS systems need to assess integration capabilities carefully rather than assuming modern systems will connect easily.
Key questions to ask any vendor during evaluation include:
- Which digital imaging scanners does the LIS natively integrate with?
- What data formats are used for image management and are they DICOM-compliant?
- How are AI analysis results returned to the LIS and surfaced to the pathologist?
- What EHR systems does the platform have validated integrations with?
Workforce and Quality Implications
The workforce implications of digital pathology are significant and complex. On one hand, digital tools enable remote review and subspecialty consultation that can extend the reach of pathology expertise, allowing a community hospital lab to access subspecialty opinion for rare or complex cases. On the other hand, the transition to digital workflows requires training and adjustment from pathologists and lab staff who have worked in analog environments.
Quality management in digital pathology labs is also an area that is still developing best practices. Labs implementing digital pathology need systematic answers to questions such as:
- How do you validate that a digital scanning process is producing images with sufficient quality for diagnosis?
- How do you monitor for scanner performance degradation over time?
- How do you handle cases where image quality is insufficient and glass slide review is needed?
The labs that are building this infrastructure now are not just improving their own operations. They are contributing to a healthcare system that can deliver more consistent, reliable diagnostic results across a wider range of settings.